- A company that meets the needs of people around the world through superior products and services
Products List

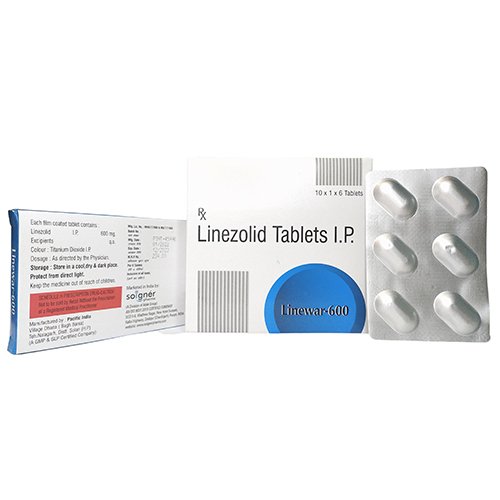
Linewar contains the formulation of Linezolid tablets, which works as an antibiotic to treat severe bacterial infection. This medicine is highly effective against bacteria that occur in the lungs and can occur in other internal organs of the body. Linezolid belongs to the family of antibacterials called oxazolidinones and works by removing bacteria which is the main reason behind infection. This drug makes the body more active and should be taken as per the prescription given by the doctor.
PRECAUTIONS FOR LINEZOLID TABLETS
One should not skip taking this medicine, take it daily before or after a meal. The consumer should Inform the doctor about the medicines they already take to avoid the symptoms of allergy. For receiving better results Take this medicine daily at a fixed time.
SIDE EFFECTS OF LINEZOLID TABLETS
Linezolid tablets do not work for cold or any other viral infection. This medication can cause some side effects but if they are in minor form and the body is adjusted to take Linezolid tablets then it can be neglected. But it can have some mild side effects in some cases if the condition gets worse and takes consultation from the doctor.
- Headache
- Decreased blood cells
- Nausea
- Diarrhea
- Vomiting
CONTACT FOR LINEZOLID TABLETS MANUFACTURING AND SUPPLY
Soigner Pharma is a well-known company in the pharmaceutical market and deals as a supplier and manufacturer of Linezolid tablets, the company also provides the best PCD pharma franchise of Linezolid tablets and other pharmaceutical products in PAN India. Being a Top third-party manufacturer we provide the best deals in all the drug ranges including tablets, capsules, injections, syrups. etc. We have an ISO certification and give more emphasis on manufacturing premium quality products while maintaining the national and international standards.
- We manufacture the product as per the guidelines of WHO and GMP in order to match the international standard of the product.
- The company has professional staff who are well-qualified in the pharma field and have long experience.
- Our units are equipped with hi-tech machinery which is able to manufacture the bulk of stock of high-quality products.
- All pharma products are approved by DCGI and FSSAI.
| Brand Name | LINEWAR 600 |
|---|---|
| Composition | LINEZOLID 600 MG |
| Pack Type | ALU-ALU |
| Pack Size | 10X1X4 |

 Get a Quote
Get a Quote